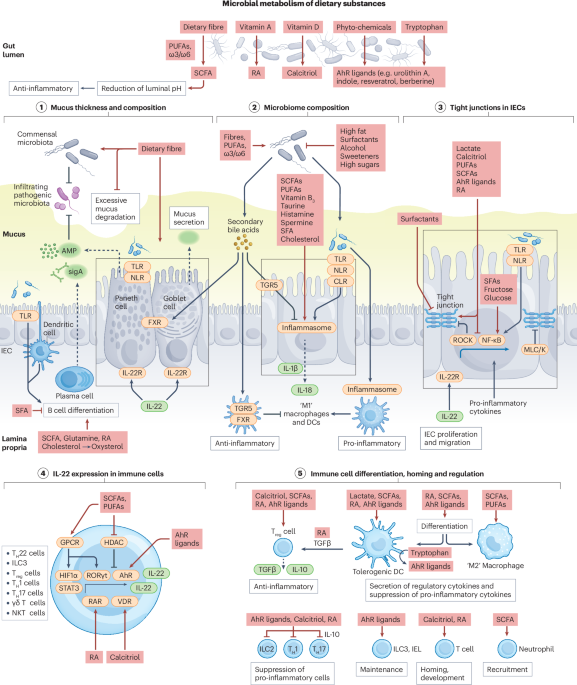
The gut microbiome connects nutrition and human health
GBD 2021 Risk Factors Collaborators. Global burden and strength of evidence for 88 risk factors in 204 countries and 811 subnational locations, 1990-2021: a systematic analysis for the Global Burden of Disease Study 2021. Lancet 403, 2162–2203 (2024).
Google Scholar
Willett, W. et al. Food in the anthropocene: the EAT-Lancet Commission on healthy diets from sustainable food systems. Lancet 393, 447–492 (2019).
Google Scholar
Mozaffarian, D., Callahan, E. A., Glickman, D. & Maitin-Shepard, M. Developing a national nutrition policy strategy to advance cardiometabolic health and health equity. Cell Metab. 36, 651–654 (2024).
Google Scholar
EFSA Panel on Dietetic Products, Nutrition, and Allergies (NDA) Scientific opinion on establishing food‐based dietary guidelines. EFSA J. 8, 1460 (2010).
Gkouskou, K. K. et al. A genomics perspective of personalized prevention and management of obesity. Hum. Genomics 18, 4 (2024).
Google Scholar
Sanz, Y. et al. Towards microbiome-informed dietary recommendations for promoting metabolic and mental health: opinion papers of the MyNewGut project. Clin. Nutr. 37, 2191–2197 (2018).
Google Scholar
Chadaideh, K. S. & Carmody, R. N. Host-microbial interactions in the metabolism of different dietary fats. Cell Metab. 33, 857–872 (2021).
Google Scholar
Perler, B. K., Friedman, E. S. & Wu, G. D. The role of the gut microbiota in the relationship between diet and human health. Annu. Rev. Physiol. 85, 449–468 (2023).
Google Scholar
Wolter, M. et al. Leveraging diet to engineer the gut microbiome. Nat. Rev. Gastroenterol. Hepatol. 18, 885–902 (2021).
Google Scholar
Fan, Y. & Pedersen, O. Gut microbiota in human metabolic health and disease. Nat. Rev. Microbiol. 19, 55–71 (2021).
Google Scholar
Carmody, R. N., Varady, K. & Turnbaugh, P. J. Digesting the complex metabolic effects of diet on the host and microbiome. Cell 187, 3857–3876 (2024).
Google Scholar
Kolodziejczyk, A. A., Zheng, D. & Elinav, E. Diet-microbiota interactions and personalized nutrition. Nat. Rev. Microbiol. 17, 742–753 (2019).
Google Scholar
Wingrove, K., Lawrence, M. A. & McNaughton, S. A. A systematic review of the methods used to assess and report dietary patterns. Front. Nutr. 9, 892351 (2022).
Google Scholar
Carson, T. L. et al. Rationale and study protocol for a randomized controlled feeding study to determine the structural- and functional-level effects of diet-specific interventions on the gut microbiota of non-Hispanic black and white adults. Contemp. Clin. Trials 123, 106968 (2022).
Google Scholar
Bowyer, R. C. E. et al. Use of dietary indices to control for diet in human gut microbiota studies. Microbiome 6, 77 (2018).
Google Scholar
Xiao, C. et al. Associations of dietary diversity with the gut microbiome, fecal metabolites, and host metabolism: results from 2 prospective Chinese cohorts. Am. J. Clin. Nutr. 116, 1049–1058 (2022).
Google Scholar
Asnicar, F. et al. Microbiome connections with host metabolism and habitual diet from 1,098 deeply phenotyped individuals. Nat. Med. 27, 321–332 (2021).
Google Scholar
Choi, Y., Hoops, S. L., Thoma, C. J. & Johnson, A. J. A guide to dietary pattern–microbiome data integration. J. Nutr. 152, 1187–1199 (2022).
Google Scholar
Cotillard, A. et al. A posteriori dietary patterns better explain variations of the gut microbiome than individual markers in the American Gut Project. Am. J. Clin. Nutr. 115, 432–443 (2022).
Google Scholar
Kase, B. E. et al. The development and evaluation of a literature-based dietary index for gut microbiota. Nutrients 16, 1045 (2024).
Google Scholar
Tagliamonte, S. et al. Relationships between diet and gut microbiome in an Italian and Dutch cohort: does the dietary protein to fiber ratio play a role? Eur. J. Nutr. 63, 741–750 (2024).
Google Scholar
Wu, G. D. et al. Linking long-term dietary patterns with gut microbial enterotypes. Science 334, 105–108 (2011).
Google Scholar
David, L. A. et al. Diet rapidly and reproducibly alters the human gut microbiome. Nature 505, 559–563 (2014).
Google Scholar
O’Keefe, S. J. D. et al. Fat, fibre and cancer risk in African Americans and rural Africans. Nat. Commun. 6, 6342 (2015).
Google Scholar
Ghosh, T. S. et al. Mediterranean diet intervention alters the gut microbiome in older people reducing frailty and improving health status: the NU-AGE 1-year dietary intervention across five European countries. Gut 69, 1218–1228 (2020).
Google Scholar
Fragiadakis, G. K. et al. Long-term dietary intervention reveals resilience of the gut microbiota despite changes in diet and weight. Am. J. Clin. Nutr. 111, 1127–1136 (2020).
Google Scholar
Muralidharan, J. et al. Effect on gut microbiota of a 1-y lifestyle intervention with Mediterranean diet compared with energy-reduced Mediterranean diet and physical activity promotion: PREDIMED-plus study. Am. J. Clin. Nutr. 114, 1148–1158 (2021).
Google Scholar
Bermingham, K. M. et al. Effects of a personalized nutrition program on cardiometabolic health: a randomized controlled trial. Nat. Med. 30, 1888–1897 (2024).
Google Scholar
Wan, Y. et al. Effects of dietary fat on gut microbiota and faecal metabolites, and their relationship with cardiometabolic risk factors: a 6-month randomised controlled-feeding trial. Gut 68, 1417–1429 (2019).
Google Scholar
Vangay, P. et al. US immigration westernizes the human gut microbiome. Cell 175, 962–972.e10 (2018).
Google Scholar
Gutierrez Lopez, D. E., Lashinger, L. M., Weinstock, G. M. & Bray, M. S. Circadian rhythms and the gut microbiome synchronize the host’s metabolic response to diet. Cell Metab. 33, 873–887 (2021).
Google Scholar
Lotti, S., Dinu, M., Colombini, B., Amedei, A. & Sofi, F. Circadian rhythms, gut microbiota, and diet: possible implications for health. Nutr. Metab. Cardiovasc. Dis. 33, 1490–1500 (2023).
Google Scholar
Ratiner, K., Shapiro, H., Goldenberg, K. & Elinav, E. Time-limited diets and the gut microbiota in cardiometabolic disease. J. Diabetes 14, 377–393 (2022).
Google Scholar
Bermingham, K. M. et al. Exploring the relationship between social jetlag with gut microbial composition, diet and cardiometabolic health, in the ZOE PREDICT 1 cohort. Eur. J. Nutr. 62, 3135–3147 (2023).
Google Scholar
Zhernakova, A. et al. Population-based metagenomics analysis reveals markers for gut microbiome composition and diversity. Science 352, 565–569 (2016).
Google Scholar
Falony, G. et al. Population-level analysis of gut microbiome variation. Science 352, 560–564 (2016).
Google Scholar
Frankenfeld, C. L. et al. The gut microbiome is associated with circulating dietary biomarkers of fruit and vegetable intake in a multiethnic cohort. J. Acad. Nutr. Diet. 122, 78–98 (2022).
Google Scholar
Manor, O. et al. Health and disease markers correlate with gut microbiome composition across thousands of people. Nat. Commun. 11, 5206 (2020).
Google Scholar
Partula, V. et al. Associations between usual diet and gut microbiota composition: results from the Milieu Intérieur cross-sectional study. Am. J. Clin. Nutr. 109, 1472–1483 (2019).
Google Scholar
Maukonen, M. et al. Associations of plant-based foods, red and processed meat, and dairy with gut microbiome in Finnish adults. Eur. J. Nutr. 63, 2247–2260 (2024).
Google Scholar
Koponen, K. K. et al. Associations of healthy food choices with gut microbiota profiles. Am. J. Clin. Nutr. 114, 605–616 (2021).
Google Scholar
Yu, D. et al. Long-term diet quality is associated with gut microbiome diversity and composition among urban Chinese adults. Am. J. Clin. Nutr. 113, 684–694 (2021).
Google Scholar
Borrello, K. et al. Dietary intake mediates ethnic differences in gut microbial composition. Nutrients 14, 660 (2022).
Google Scholar
Latorre-Pérez, A. et al. The Spanish gut microbiome reveals links between microorganisms and Mediterranean diet. Sci. Rep. 11, 21602 (2021).
Google Scholar
Houttu, V. et al. Physical activity and dietary composition relate to differences in gut microbial patterns in a multi-ethnic cohort – the HELIUS study. Metabolites 11, 858 (2021).
Google Scholar
Le Roy, C. I. et al. Yoghurt consumption is associated with changes in the composition of the human gut microbiome and metabolome. BMC Microbiol. 22, 39 (2022).
Google Scholar
Jiang, Z. et al. Dietary fruit and vegetable intake, gut microbiota, and type 2 diabetes: results from two large human cohort studies. BMC Med. 18, 371 (2020).
Google Scholar
Aasmets, O., Krigul, K. L., Lüll, K., Metspalu, A. & Org, E. Gut metagenome associations with extensive digital health data in a volunteer-based Estonian microbiome cohort. Nat. Commun. 13, 869 (2022).
Google Scholar
Cotillard, A. et al. Dietary intervention impact on gut microbial gene richness. Nature 500, 585–588 (2013).
Google Scholar
Miller, V. et al. Global dietary quality in 185 countries from 1990 to 2018 show wide differences by nation, age, education, and urbanicity. Nat. Food 3, 694–702 (2022).
Google Scholar
Jardon, K. M., Canfora, E. E., Goossens, G. H. & Blaak, E. E. Dietary macronutrients and the gut microbiome: a precision nutrition approach to improve cardiometabolic health. Gut 71, 1214–1226 (2022).
Google Scholar
Arnone, D. et al. Sugars and gastrointestinal health. Clin. Gastroenterol. Hepatol. 20, 1912–1924.e7 (2022).
Google Scholar
Fernández-Bañares, F. Carbohydrate maldigestion and intolerance. Nutrients 14, 1923 (2022).
Google Scholar
Blachier, F. et al. High-protein diets for weight management: interactions with the intestinal microbiota and consequences for gut health. A position paper by the My New Gut study group. Clin. Nutr. 38, 1012–1022 (2019).
Google Scholar
Bartlett, A. & Kleiner, M. Dietary protein and the intestinal microbiota: an understudied relationship. iScience 25, 105313 (2022).
Google Scholar
Fassarella, M. et al. Gut microbiome stability and resilience: elucidating the response to perturbations in order to modulate gut health. Gut 70, 595–605 (2021).
Google Scholar
Li, D. et al. Gut microbiota-derived metabolite trimethylamine-N-oxide and multiple health outcomes: an umbrella review and updated meta-analysis. Am. J. Clin. Nutr. 116, 230–243 (2022).
Google Scholar
Wolters, M. et al. Dietary fat, the gut microbiota, and metabolic health – a systematic review conducted within the MyNewGut project. Clin. Nutr. 38, 2504–2520 (2019).
Google Scholar
Lemons, J. M. S. & Liu, L. Chewing the fat with microbes: lipid crosstalk in the gut. Nutrients 14, 573 (2022).
Google Scholar
Natividad, J. M. et al. Bilophila wadsworthia aggravates high fat diet induced metabolic dysfunctions in mice. Nat. Commun. 9, 2802 (2018).
Google Scholar
Collins, S. L., Stine, J. G., Bisanz, J. E., Okafor, C. D. & Patterson, A. D. Bile acids and the gut microbiota: metabolic interactions and impacts on disease. Nat. Rev. Microbiol. 21, 236–247 (2023).
Google Scholar
Pham, V. T., Dold, S., Rehman, A., Bird, J. K. & Steinert, R. E. Vitamins, the gut microbiome and gastrointestinal health in humans. Nutr. Res. 95, 35–53 (2021).
Google Scholar
Wilck, N. et al. Salt-responsive gut commensal modulates TH17 axis and disease. Nature 551, 585–589 (2017).
Google Scholar
Musiol, S. et al. The impact of high-salt diet on asthma in humans and mice: effect on specific T-cell signatures and microbiome. Allergy 79, 1844–1857 (2024).
Google Scholar
Spiga, L. et al. Iron acquisition by a commensal bacterium modifies host nutritional immunity during Salmonella infection. Cell Host Microbe 31, 1639–1654.e10 (2023).
Google Scholar
Martin, O. C. B. et al. Haem iron reshapes colonic luminal environment: impact on mucosal homeostasis and microbiome through aldehyde formation. Microbiome 7, 72 (2019).
Google Scholar
Mervant, L. et al. Urinary metabolome analysis reveals potential microbiota alteration and electrophilic burden induced by high red meat diet: results from the French NutriNet-Santé cohort and an in vivo intervention study in rats. Mol. Nutr. Food Res. 67, e2200432 (2023).
Google Scholar
Rowland, I. et al. Gut microbiota functions: metabolism of nutrients and other food components. Eur. J. Nutr. 57, 1–24 (2018).
Google Scholar
Pereira, Q. C. et al. Polyphenolic compounds: orchestrating intestinal microbiota harmony during aging. Nutrients 16, 1066 (2024).
Google Scholar
Meijnikman, A. S. et al. Microbiome-derived ethanol in nonalcoholic fatty liver disease. Nat. Med. 28, 2100–2106 (2022).
Google Scholar
Srour, B. et al. Ultra-processed foods and human health: from epidemiological evidence to mechanistic insights. Lancet Gastroenterol. Hepatol. 7, 1128–1140 (2022).
Google Scholar
Shalon, D. et al. Profiling the human intestinal environment under physiological conditions. Nature 617, 581–591 (2023).
Google Scholar
Quinn, R. A. et al. Global chemical effects of the microbiome include new bile-acid conjugations. Nature 579, 123–129 (2020).
Google Scholar
Di Vincenzo, F., Del Gaudio, A., Petito, V., Lopetuso, L. R. & Scaldaferri, F. Gut microbiota, intestinal permeability, and systemic inflammation: a narrative review. Intern. Emerg. Med. 19, 275–293 (2024).
Google Scholar
Chae, Y.-R., Lee, Y. R., Kim, Y.-S. & Park, H.-Y. Diet-induced gut dysbiosis and leaky gut syndrome. J. Microbiol. Biotechnol. 34, 747–756 (2024).
Google Scholar
Ito, H. et al. Soluble fiber viscosity affects both goblet cell number and small intestine mucin secretion in rats. J. Nutr. 139, 1640–1647 (2009).
Google Scholar
Desai, M. S. et al. A dietary fiber-deprived gut microbiota degrades the colonic mucus barrier and enhances pathogen susceptibility. Cell 167, 1339–1353.e21 (2016).
Google Scholar
Belzer, C. Nutritional strategies for mucosal health: the interplay between microbes and mucin glycans. Trends Microbiol. 30, 13–21 (2022).
Google Scholar
Mukherjee, S. & Hooper, L. V. Antimicrobial defense of the intestine. Immunity 42, 28–39 (2015).
Google Scholar
Keir, M., Yi, T., Lu, T. & Ghilardi, N. The role of IL-22 in intestinal health and disease. J. Exp. Med. 217, e20192195 (2020).
Google Scholar
Feng, Z., Sun, R., Cong, Y. & Liu, Z. Critical roles of G protein-coupled receptors in regulating intestinal homeostasis and inflammatory bowel disease. Mucosal Immunol. 15, 819–828 (2022).
Google Scholar
Stockinger, B., Shah, K. & Wincent, E. AHR in the intestinal microenvironment: safeguarding barrier function. Nat. Rev. Gastroenterol. Hepatol. 18, 559–570 (2021).
Google Scholar
Kahalehili, H. M. et al. Dietary indole-3-carbinol activates AhR in the gut, alters Th17-microbe interactions, and exacerbates insulitis in NOD mice. Front. Immunol. 11, 606441 (2020).
Google Scholar
Song, Y. et al. Molecular and structural basis of interactions of vitamin D3 hydroxyderivatives with aryl hydrocarbon receptor (AhR): an integrated experimental and computational study. Int. J. Biol. Macromol. 209, 1111–1123 (2022).
Google Scholar
Bora, S. A., Kennett, M. J., Smith, P. B., Patterson, A. D. & Cantorna, M. T. The gut microbiota regulates endocrine vitamin D metabolism through fibroblast growth factor 23. Front. Immunol. 9, 408 (2018).
Google Scholar
Aoki, R., Aoki-Yoshida, A., Suzuki, C. & Takayama, Y. Indole-3-pyruvic acid, an aryl hydrocarbon receptor activator, suppresses experimental colitis in mice. J. Immunol. 201, 3683–3693 (2018).
Google Scholar
Woo, V. et al. Commensal segmented filamentous bacteria-derived retinoic acid primes host defense to intestinal infection. Cell Host Microbe 29, 1744–1756.e5 (2021).
Google Scholar
Han, X. et al. Dietary regulation of the SIgA–gut microbiota interaction. Crit. Rev. Food Sci. Nutr. 63, 6379–6392 (2023).
Google Scholar
Shin, S. Y. et al. Compositional changes in the gut microbiota of responders and non-responders to probiotic treatment among patients with diarrhea-predominant irritable bowel syndrome: a post hoc analysis of a randomized clinical trial. J. Neurogastroenterol. Motil. 28, 642–654 (2022).
Google Scholar
Yu, M. et al. Aryl hydrocarbon receptor activation modulates intestinal epithelial barrier function by maintaining tight junction integrity. Int. J. Biol. Sci. 14, 69–77 (2018).
Google Scholar
Feng, W. et al. Sodium butyrate attenuates diarrhea in weaned piglets and promotes tight junction protein expression in colon in a GPR109A-dependent manner. Cell. Physiol. Biochem. 47, 1617–1629 (2018).
Google Scholar
Li, X., Yao, Z., Qian, J., Li, H. & Li, H. Lactate protects intestinal epithelial barrier function from dextran sulfate sodium-induced damage by GPR81 signaling. Nutrients 16, 582 (2024).
Google Scholar
Venkatesh, M. et al. Symbiotic bacterial metabolites regulate gastrointestinal barrier function via the xenobiotic sensor PXR and toll-like receptor 4. Immunity 41, 296–310 (2014).
Google Scholar
He, C. et al. Vitamin A inhibits the action of LPS on the intestinal epithelial barrier function and tight junction proteins. Food Funct. 10, 1235–1242 (2019).
Google Scholar
Cho, Y.-E. et al. Fructose promotes leaky gut, endotoxemia, and liver fibrosis through ethanol-inducible cytochrome P450-2E1-mediated oxidative and nitrative stress. Hepatology 73, 2180–2195 (2021).
Google Scholar
Rohr, M. W., Narasimhulu, C. A., Rudeski-Rohr, T. A. & Parthasarathy, S. Negative effects of a high-fat diet on intestinal permeability: a review. Adv. Nutr. 11, 77–91 (2020).
Google Scholar
Chassaing, B. et al. Dietary emulsifiers impact the mouse gut microbiota promoting colitis and metabolic syndrome. Nature 519, 92–96 (2015).
Google Scholar
Siracusa, F. et al. Short-term dietary changes can result in mucosal and systemic immune depression. Nat. Immunol. 24, 1473–1486 (2023).
Google Scholar
Tan, J. et al. Your regulatory T cells are what you eat: how diet and gut microbiota affect regulatory T cell development. Front. Nutr. 9, 878382 (2022).
Google Scholar
Bakdash, G., Vogelpoel, L. T. C., van Capel, T. M. M., Kapsenberg, M. L. & de Jong, E. C. Retinoic acid primes human dendritic cells to induce gut-homing, IL-10-producing regulatory T cells. Mucosal Immunol. 8, 265–278 (2015).
Google Scholar
Xie, L., Alam, M. J., Marques, F. Z. & Mackay, C. R. A major mechanism for immunomodulation: dietary fibres and acid metabolites. Semin. Immunol. 66, 101737 (2023).
Google Scholar
Kayama, H. & Takeda, K. Emerging roles of host and microbial bioactive lipids in inflammatory bowel diseases. Eur. J. Immunol. 53, e2249866 (2023).
Google Scholar
Urry, Z. et al. The role of 1α,25-dihydroxyvitamin D3 and cytokines in the promotion of distinct Foxp3+ and IL-10+ CD4+ T cells. Eur. J. Immunol. 42, 2697–2708 (2012).
Google Scholar
Quintana, F. J. et al. Control of Treg and TH17 cell differentiation by the aryl hydrocarbon receptor. Nature 453, 65–71 (2008).
Google Scholar
Kelly, B. & Pearce, E. L. Amino assets: how amino acids support immunity. Cell Metab. 32, 154–175 (2020).
Google Scholar
Bauché, D. & Marie, J. C. Transforming growth factor β: a master regulator of the gut microbiota and immune cell interactions. Clin. Transl. Immunol. 6, e136 (2017).
Google Scholar
Elias, K. M. et al. Retinoic acid inhibits Th17 polarization and enhances FoxP3 expression through a Stat-3/Stat-5 independent signaling pathway. Blood 111, 1013–1020 (2008).
Google Scholar
Paudel, S., Mishra, N. & Agarwal, R. Phytochemicals as immunomodulatory molecules in cancer therapeutics. Pharmaceuticals 16, 1652 (2023).
Google Scholar
Rodrigues, H. G., Takeo Sato, F., Curi, R. & Vinolo, M. A. R. Fatty acids as modulators of neutrophil recruitment, function and survival. Eur. J. Pharmacol. 785, 50–58 (2016).
Google Scholar
Schulthess, J. et al. The short chain fatty acid butyrate imprints an antimicrobial program in macrophages. Immunity 50, 432–445.e7 (2019).
Google Scholar
Nastasi, C. et al. Butyrate and propionate inhibit antigen-specific CD8+ T cell activation by suppressing IL-12 production by antigen-presenting cells. Sci. Rep. 7, 14516 (2017).
Google Scholar
Li, S., Bostick, J. W. & Zhou, L. Regulation of innate lymphoid cells by aryl hydrocarbon receptor. Front. Immunol. 8, 1909 (2017).
Google Scholar
Li, Y. et al. Exogenous stimuli maintain intraepithelial lymphocytes via aryl hydrocarbon receptor activation. Cell 147, 629–640 (2011).
Google Scholar
Inagaki, T. et al. Regulation of antibacterial defense in the small intestine by the nuclear bile acid receptor. Proc. Natl Acad. Sci. USA 103, 3920–3925 (2006).
Google Scholar
Gadaleta, R. M. et al. Farnesoid X receptor activation inhibits inflammation and preserves the intestinal barrier in inflammatory bowel disease. Gut 60, 463–472 (2011).
Google Scholar
Thibaut, M. M. & Bindels, L. B. Crosstalk between bile acid-activated receptors and microbiome in entero-hepatic inflammation. Trends Mol. Med. 28, 223–236 (2022).
Google Scholar
Zmora, N., Levy, M., Pevsner-Fishcer, M. & Elinav, E. Inflammasomes and intestinal inflammation. Mucosal Immunol. 10, 865–883 (2017).
Google Scholar
Swanson, K. V., Deng, M. & Ting, J. P.-Y. The NLRP3 inflammasome: molecular activation and regulation to therapeutics. Nat. Rev. Immunol. 19, 477–489 (2019).
Google Scholar
Zheng, D. et al. Epithelial Nlrp10 inflammasome mediates protection against intestinal autoinflammation. Nat. Immunol. 24, 585–594 (2023).
Google Scholar
Macia, L. et al. Metabolite-sensing receptors GPR43 and GPR109A facilitate dietary fibre-induced gut homeostasis through regulation of the inflammasome. Nat. Commun. 6, 6734 (2015).
Google Scholar
Wen, H. et al. Fatty acid-induced NLRP3-ASC inflammasome activation interferes with insulin signaling. Nat. Immunol. 12, 408–415 (2011).
Google Scholar
Yan, Y. et al. Omega-3 fatty acids prevent inflammation and metabolic disorder through inhibition of NLRP3 inflammasome activation. Immunity 38, 1154–1163 (2013).
Google Scholar
Schneider, E., O’Riordan, K. J., Clarke, G. & Cryan, J. F. Feeding gut microbes to nourish the brain: unravelling the diet-microbiota–gut–brain axis. Nat. Metab. 6, 1454–1478 (2024).
Google Scholar
Knox, E. G. et al. Microbial-derived metabolites induce actin cytoskeletal rearrangement and protect blood–brain barrier function. iScience 25, 105648 (2022).
Google Scholar
O’Riordan, K. J. et al. Short chain fatty acids: microbial metabolites for gut–brain axis signalling. Mol. Cell. Endocrinol. 546, 111572 (2022).
Google Scholar
Spichak, S. et al. Mining microbes for mental health: determining the role of microbial metabolic pathways in human brain health and disease. Neurosci. Biobehav. Rev. 125, 698–761 (2021).
Google Scholar
McGuinness, A. J. et al. A systematic review of gut microbiota composition in observational studies of major depressive disorder, bipolar disorder and schizophrenia. Mol. Psychiatry 27, 1920–1935 (2022).
Google Scholar
Nikolova, V. L. et al. Perturbations in gut microbiota composition in psychiatric disorders: a review and meta-analysis. JAMA Psychiatry 78, 1343–1354 (2021).
Google Scholar
Aslam, H. et al. Fiber intake and fiber intervention in depression and anxiety: a systematic review and meta-analysis of observational studies and randomized controlled trials. Nutr. Rev. 82, 1678–1695 (2024).
Google Scholar
Provensi, G. et al. Preventing adolescent stress-induced cognitive and microbiome changes by diet. Proc. Natl Acad. Sci. USA 116, 9644–9651 (2019).
Google Scholar
Kerman, B. E., Self, W. & Yassine, H. N. Can the gut microbiome inform the effects of omega-3 fatty acid supplementation trials on cognition? Curr. Opin. Clin. Nutr. Metab. Care 27, 116–124 (2024).
Google Scholar
Miyamoto, J. et al. Gut microbiota confers host resistance to obesity by metabolizing dietary polyunsaturated fatty acids. Nat. Commun. 10, 4007 (2019).
Google Scholar
Schverer, M. et al. Dietary phospholipids: role in cognitive processes across the lifespan. Neurosci. Biobehav. Rev. 111, 183–193 (2020).
Google Scholar
Ross, F. C. et al. Potential of dietary polyphenols for protection from age-related decline and neurodegeneration: a role for gut microbiota?. Nutr. Neurosci. 27, 1058–1076 (2024).
Google Scholar
Brickman, A. M. et al. Dietary flavanols restore hippocampal-dependent memory in older adults with lower diet quality and lower habitual flavanol consumption. Proc. Natl Acad. Sci. USA 120, e2216932120 (2023).
Google Scholar
Karbownik, M. S., Mokros, Ł. & Kowalczyk, E. Who benefits from fermented food consumption? A comparative analysis between psychiatrically Ill and psychiatrically healthy medical students. Int. J. Environ. Res. Public. Health 19, 3861 (2022).
Google Scholar
Balasubramanian, R., Schneider, E., Gunnigle, E., Cotter, P. D. & Cryan, J. F. Fermented foods: harnessing their potential to modulate the microbiota–gut–brain axis for mental health. Neurosci. Biobehav. Rev. 158, 105562 (2024).
Google Scholar
Caffrey, E. B., Sonnenburg, J. L. & Devkota, S. Our extended microbiome: the human-relevant metabolites and biology of fermented foods. Cell Metab. 36, 684–701 (2024).
Google Scholar
Berding, K. et al. Feed your microbes to deal with stress: a psychobiotic diet impacts microbial stability and perceived stress in a healthy adult population. Mol. Psychiatry 28, 601–610 (2023).
Google Scholar
Conn, K. A., Borsom, E. M. & Cope, E. K. Implications of microbe-derived γ-aminobutyric acid (GABA) in gut and brain barrier integrity and GABAergic signaling in Alzheimer’s disease. Gut Microbes 16, 2371950 (2024).
Google Scholar
Bermudez-Martin, P. et al. The microbial metabolite p-cresol induces autistic-like behaviors in mice by remodeling the gut microbiota. Microbiome 9, 157 (2021).
Google Scholar
Chambers, E. S. et al. Effects of targeted delivery of propionate to the human colon on appetite regulation, body weight maintenance and adiposity in overweight adults. Gut 64, 1744–1754 (2015).
Google Scholar
Zhao, L. et al. Gut bacteria selectively promoted by dietary fibers alleviate type 2 diabetes. Science 359, 1151–1156 (2018).
Google Scholar
Pedersen, M. G. B. et al. Oral lactate slows gastric emptying and suppresses appetite in young males. Clin. Nutr. 41, 517–525 (2022).
Google Scholar
Gribble, F. M. & Reimann, F. Enteroendocrine cells: chemosensors in the intestinal epithelium. Annu. Rev. Physiol. 78, 277–299 (2016).
Google Scholar
Engelstoft, M. S. et al. Seven transmembrane G protein-coupled receptor repertoire of gastric ghrelin cells. Mol. Metab. 2, 376–392 (2013).
Google Scholar
Fukumoto, S. et al. Short-chain fatty acids stimulate colonic transit via intraluminal 5-HT release in rats. Am. J. Physiol. Regul. Integr. Comp. Physiol. 284, R1269–R1276 (2003).
Google Scholar
De Vadder, F. et al. Microbiota-generated metabolites promote metabolic benefits via gut–brain neural circuits. Cell 156, 84–96 (2014).
Google Scholar
Marin, E. et al. Human tolerogenic dendritic cells regulate immune responses through lactate synthesis. Cell Metab. 30, 1075–1090.e8 (2019).
Google Scholar
Luo, Y. et al. Effects of lactate in immunosuppression and inflammation: progress and prospects. Int. Rev. Immunol. 41, 19–29 (2022).
Google Scholar
Jocken, J. W. E. et al. Short-chain fatty acids differentially affect intracellular lipolysis in a human white adipocyte model. Front. Endocrinol. 8, 372 (2017).
Google Scholar
Canfora, E. E. et al. Colonic infusions of short-chain fatty acid mixtures promote energy metabolism in overweight/obese men: a randomized crossover trial. Sci. Rep. 7, 2360 (2017).
Google Scholar
Hu, J. et al. The roles of GRP81 as a metabolic sensor and inflammatory mediator. J. Cell. Physiol. 235, 8938–8950 (2020).
Google Scholar
Reddy, A. et al. Monocarboxylate transporters facilitate succinate uptake into brown adipocytes. Nat. Metab. 6, 567–577 (2024).
Google Scholar
Kimura, I. et al. The gut microbiota suppresses insulin-mediated fat accumulation via the short-chain fatty acid receptor GPR43. Nat. Commun. 4, 1829 (2013).
Google Scholar
Villanueva-Carmona, T. et al. SUCNR1 signaling in adipocytes controls energy metabolism by modulating circadian clock and leptin expression. Cell Metab. 35, 601–619.e10 (2023).
Google Scholar
Müller, M. et al. Circulating but not faecal short-chain fatty acids are related to insulin sensitivity, lipolysis and GLP-1 concentrations in humans. Sci. Rep. 9, 12515 (2019).
Google Scholar
Frost, G. et al. The short-chain fatty acid acetate reduces appetite via a central homeostatic mechanism. Nat. Commun. 5, 3611 (2014).
Google Scholar
Perry, R. J. et al. Acetate mediates a microbiome–brain–β-cell axis to promote metabolic syndrome. Nature 534, 213–217 (2016).
Google Scholar
Wang, K. et al. Parabacteroides distasonis alleviates obesity and metabolic dysfunctions via production of succinate and secondary bile acids. Cell Rep. 26, 222–235.e5 (2019).
Google Scholar
Osuna-Prieto, F. J. et al. Elevated plasma succinate levels are linked to higher cardiovascular disease risk factors in young adults. Cardiovasc. Diabetol. 20, 151 (2021).
Google Scholar
Serena, C. et al. Elevated circulating levels of succinate in human obesity are linked to specific gut microbiota. ISME J. 12, 1642–1657 (2018).
Google Scholar
Ding, Y. et al. Oral supplementation of gut microbial metabolite indole-3-acetate alleviates diet-induced steatosis and inflammation in mice. eLife 12, RP87458 (2024).
Google Scholar
Min, B. H. et al. Gut microbiota-derived indole compounds attenuate metabolic dysfunction-associated steatotic liver disease by improving fat metabolism and inflammation. Gut Microbes 16, 2307568 (2024).
Google Scholar
Wang, Z. et al. Gut microbiota and blood metabolites related to fiber intake and type 2 diabetes. Circ. Res. 134, 842–854 (2024).
Google Scholar
Dodd, D. et al. A gut bacterial pathway metabolizes aromatic amino acids into nine circulating metabolites. Nature 551, 648–652 (2017).
Google Scholar
Lu, C. et al. Indoxyl sulfate in atherosclerosis. Toxicol. Lett. 383, 204–212 (2023).
Google Scholar
Zhu, Y. et al. Two distinct gut microbial pathways contribute to meta-organismal production of phenylacetylglutamine with links to cardiovascular disease. Cell Host Microbe 31, 18–32.e9 (2023).
Google Scholar
Romano, K. A. et al. Gut microbiota-generated phenylacetylglutamine and heart failure. Circ. Heart Fail. 16, e009972 (2023).
Google Scholar
Han, H. et al. p-Cresyl sulfate aggravates cardiac dysfunction associated with chronic kidney disease by enhancing apoptosis of cardiomyocytes. J. Am. Heart Assoc. 4, e001852 (2015).
Google Scholar
Kok, C. R., Rose, D. & Hutkins, R. Predicting personalized responses to dietary fiber interventions: opportunities for modulation of the gut microbiome to improve health. Annu. Rev. Food Sci. Technol. 14, 157–182 (2023).
Google Scholar
Pedersen, H. K. et al. Human gut microbes impact host serum metabolome and insulin sensitivity. Nature 535, 376–381 (2016).
Google Scholar
Wang, T. J. et al. Metabolite profiles and the risk of developing diabetes. Nat. Med. 17, 448–453 (2011).
Google Scholar
Fromentin, S. et al. Microbiome and metabolome features of the cardiometabolic disease spectrum. Nat. Med. 28, 303–314 (2022).
Google Scholar
Li, T.-T. et al. Microbiota metabolism of intestinal amino acids impacts host nutrient homeostasis and physiology. Cell Host Microbe 32, 661–675.e10 (2024).
Google Scholar
Choi, B. H., Hyun, S. & Koo, S.-H. The role of BCAA metabolism in metabolic health and disease. Exp. Mol. Med. 56, 1552–1559 (2024).
Google Scholar
Rigamonti, A. E. et al. The appetite-suppressant and GLP-1-stimulating effects of whey proteins in obese subjects are associated with increased circulating levels of specific amino acids. Nutrients 12, 775 (2020).
Google Scholar
Magkos, F. The role of dietary protein in obesity. Rev. Endocr. Metab. Disord. 21, 329–340 (2020).
Google Scholar
Kenny, D. J. et al. Cholesterol metabolism by uncultured human gut bacteria influences host cholesterol level. Cell Host Microbe 28, 245–257.e6 (2020).
Google Scholar
Romaní-Pérez, M. et al. Holdemanella biformis improves glucose tolerance and regulates GLP-1 signaling in obese mice. FASEB J. 35, e21734 (2021).
Google Scholar
Takeuchi, T. et al. Fatty acid overproduction by gut commensal microbiota exacerbates obesity. Cell Metab. 35, 361–375.e9 (2023).
Google Scholar
Wei, W. et al. Parabacteroides distasonis uses dietary inulin to suppress NASH via its metabolite pentadecanoic acid. Nat. Microbiol. 8, 1534–1548 (2023).
Google Scholar
Johnson, E. L. et al. Sphingolipids produced by gut bacteria enter host metabolic pathways impacting ceramide levels. Nat. Commun. 11, 2471 (2020).
Google Scholar
Liu, C. et al. Gut commensal Christensenella minuta modulates host metabolism via acylated secondary bile acids. Nat. Microbiol. 9, 434–450 (2024).
Google Scholar
Kuhre, R. E. et al. Bile acids are important direct and indirect regulators of the secretion of appetite- and metabolism-regulating hormones from the gut and pancreas. Mol. Metab. 11, 84–95 (2018).
Google Scholar
Zheng, X. et al. Hyocholic acid species improve glucose homeostasis through a distinct TGR5 and FXR signaling mechanism. Cell Metab. 33, 791–803.e7 (2021).
Google Scholar
Browning, M. G., Pessoa, B. M., Khoraki, J. & Campos, G. M. Changes in bile acid metabolism, transport, and signaling as central drivers for metabolic improvements after bariatric surgery. Curr. Obes. Rep. 8, 175–184 (2019).
Google Scholar
Makki, K. et al. 6α-hydroxylated bile acids mediate TGR5 signalling to improve glucose metabolism upon dietary fiber supplementation in mice. Gut 72, 314–324 (2023).
Google Scholar
Hang, S. et al. Bile acid metabolites control TH17 and Treg cell differentiation. Nature 576, 143–148 (2019).
Google Scholar
Wahlström, A. et al. Production of deoxycholic acid by low-abundant microbial species is associated with impaired glucose metabolism. Nat. Commun. 15, 4276 (2024).
Google Scholar
Guzior, D. V. et al. Bile salt hydrolase acyltransferase activity expands bile acid diversity. Nature 626, 852–858 (2024).
Google Scholar
Wu, H. et al. The gut microbiota in prediabetes and diabetes: a population-based cross-sectional study. Cell Metab. 32, 379–390.e3 (2020).
Google Scholar
Belda, E. et al. Impairment of gut microbial biotin metabolism and host biotin status in severe obesity: effect of biotin and prebiotic supplementation on improved metabolism. Gut 71, 2463–2480 (2022).
Google Scholar
Sun, Y. et al. Parabacteroides distasonis ameliorates insulin resistance via activation of intestinal GPR109a. Nat. Commun. 14, 7740 (2023).
Google Scholar
Man, A. W. C., Zhou, Y., Xia, N. & Li, H. Involvement of gut microbiota, microbial metabolites and interaction with polyphenol in host immunometabolism. Nutrients 12, 3054 (2020).
Google Scholar
Yoon, H. S. et al. Akkermansia muciniphila secretes a glucagon-like peptide-1-inducing protein that improves glucose homeostasis and ameliorates metabolic disease in mice. Nat. Microbiol. 6, 563–573 (2021).
Google Scholar
Grasset, E. et al. A specific gut microbiota dysbiosis of type 2 diabetic mice induces GLP-1 resistance through an enteric NO-dependent and gut–brain axis mechanism. Cell Metab. 25, 1075–1090.e5 (2017).
Google Scholar
Liang, C. et al. Ligilactobacillus salivarius LCK11 prevents obesity by promoting PYY secretion to inhibit appetite and regulating gut microbiota in C57BL/6J mice. Mol. Nutr. Food Res. 65, e2100136 (2021).
Google Scholar
Food and Agriculture Organization. Dietary guidelines. FAO www.fao.org/nutrition/education/food-dietary-guidelines/home/en/ (2025).
Hercberg, S., Touvier, M., Salas-Salvado, J. & Group of European scientists supporting the implementation of Nutri-Score in Europe. The nutri-score nutrition label. Int. J. Vitam. Nutr. Res. 92, 147–157 (2022).
Google Scholar
Nutri-Score European Scientific Committee. Nutri-Score. Santé publique, France www.santepubliquefrance.fr/en/nutri-score (2025).
Reyes, M. et al. Development of the Chilean front-of-package food warning label. BMC Public. Health 19, 906 (2019).
Google Scholar
Srour, B. et al. Effect of a new graphically modified Nutri-Score on the objective understanding of foods’ nutrient profile and ultraprocessing: a randomised controlled trial. BMJ Nutr. Prev. Health 6, 108–118 (2023).
Google Scholar
Merten, C. et al. Editorial: exploring the need to include microbiomes into EFSA’s scientific assessments. EFSA J. 18, e18061 (2020).
Google Scholar
Garcia-Vello, P. et al. Preparing for future challenges in risk assessment in the European Union. Trends Biotechnol. 40, 1137–1140 (2022).
Google Scholar
EFSA Panel on Dietetic Products, Nutrition, and Allergies (NDA) Scientific opinion on dietary reference values for carbohydrates and dietary fibre. EFSA J. 8, 1462 (2010).
Hoge Gezonheidsraad. Voedingsaanbevelingen voor Belgie. HGR Nr. 9285. HGR www.health.belgium.be/sites/default/files/uploads/fields/fpshealth_theme_file/9285_voedingsaanbev_16122016_a5.pdf (2016).
Vorster, H. H., Badham, J. B. & Venter, C. S. An introduction to the revised food-based dietary guidelines for South Africa. S. Afr. J. Clin. Nutr. 26, S5–S12 (2013).
Dietary Guidelines Advisory Committee. Scientific report of the 2020 Dietary Guidelines Advisory Committee: advisory report to the Secretary of Agriculture and the Secretary of Health and Human Services. Dietary Guidelines for Americans www.dietaryguidelines.gov/2020-advisory-committee-report (2020).
McKeown, N. M., Fahey, G. C., Slavin, J. & van der Kamp, J.-W. Fibre intake for optimal health: how can healthcare professionals support people to reach dietary recommendations? BMJ 378, e054370 (2022).
Google Scholar
Ou, J. et al. Diet, microbiota, and microbial metabolites in colon cancer risk in rural Africans and African Americans. Am. J. Clin. Nutr. 98, 111–120 (2013).
Google Scholar
Zmora, N., Suez, J. & Elinav, E. You are what you eat: diet, health and the gut microbiota. Nat. Rev. Gastroenterol. Hepatol. 16, 35–56 (2019).
Google Scholar
Appel, L. J. et al. A clinical trial of the effects of dietary patterns on blood pressure. DASH collaborative research group. N. Engl. J. Med. 336, 1117–1124 (1997).
Google Scholar
Paeslack, N. et al. Microbiota-derived tryptophan metabolites in vascular inflammation and cardiovascular disease. Amino Acids 54, 1339–1356 (2022).
Google Scholar
Kaye, D. M. et al. Deficiency of prebiotic fiber and insufficient signaling through gut metabolite-sensing receptors leads to cardiovascular disease. Circulation 141, 1393–1403 (2020).
Google Scholar
Meslier, V. et al. Mediterranean diet intervention in overweight and obese subjects lowers plasma cholesterol and causes changes in the gut microbiome and metabolome independently of energy intake. Gut 69, 1258–1268 (2020).
Google Scholar
Shoer, S. et al. Impact of dietary interventions on pre-diabetic oral and gut microbiome, metabolites and cytokines. Nat. Commun. 14, 5384 (2023).
Google Scholar
Dinan, T. G. et al. Feeding melancholic microbes: MyNewGut recommendations on diet and mood. Clin. Nutr. 38, 1995–2001 (2019).
Google Scholar
Morris, M. C. et al. MIND diet slows cognitive decline with aging. Alzheimers Dement. 11, 1015–1022 (2015).
Google Scholar
Agarwal, P. et al. MIND diet associated with reduced incidence and delayed progression of parkinsonism in old age. J. Nutr. Health Aging 22, 1211–1215 (2018).
Google Scholar
Erny, D. et al. Host microbiota constantly control maturation and function of microglia in the CNS. Nat. Neurosci. 18, 965–977 (2015).
Google Scholar
MahmoudianDehkordi, S. et al. Altered bile acid profile associates with cognitive impairment in Alzheimer’s disease – an emerging role for gut microbiome. Alzheimers Dement. 15, 76–92 (2019).
Google Scholar
Valls-Pedret, C. et al. Polyphenol-rich foods in the Mediterranean diet are associated with better cognitive function in elderly subjects at high cardiovascular risk. J. Alzheimers Dis. 29, 773–782 (2012).
Google Scholar
Torabynasab, K. et al. Adherence to the MIND diet is inversely associated with odds and severity of anxiety disorders: a case-control study. BMC Psychiatry 23, 330 (2023).
Google Scholar
Prince, A. C. et al. Fermentable carbohydrate restriction (low FODMAP diet) in clinical practice improves functional gastrointestinal symptoms in patients with inflammatory bowel disease. Inflamm. Bowel Dis. 22, 1129–1136 (2016).
Google Scholar
Kamphuis, J. B. J. et al. Lactose and fructo-oligosaccharides increase visceral sensitivity in mice via glycation processes, increasing mast cell density in colonic mucosa. Gastroenterology 158, 652–663.e6 (2020).
Google Scholar
Wielgosz-Grochowska, J. P., Domanski, N. & Drywień, M. E. Efficacy of an irritable bowel syndrome diet in the treatment of small intestinal bacterial overgrowth: a narrative review. Nutrients 14, 3382 (2022).
Google Scholar
Garg, P., Garg, P. K. & Bhattacharya, K. Psyllium husk positively alters gut microbiota, decreases inflammation, and has bowel-regulatory action, paving the way for physiologic management of irritable bowel syndrome. Gastroenterology 166, 545–546 (2024).
Google Scholar
Sanz, Y. Microbiome and gluten. Ann. Nutr. Metab. 67, 28–41 (2015).
Google Scholar
Caminero, A. et al. Duodenal bacterial proteolytic activity determines sensitivity to dietary antigen through protease-activated receptor-2. Nat. Commun. 10, 1198 (2019).
Google Scholar
Matera, M. & Guandalini, S. How the microbiota may affect celiac disease and what we can do. Nutrients 16, 1882 (2024).
Google Scholar
Adolfsen, K. J. et al. Improvement of a synthetic live bacterial therapeutic for phenylketonuria with biosensor-enabled enzyme engineering. Nat. Commun. 12, 6215 (2021).
Google Scholar
Bassanini, G. et al. Phenylketonuria diet promotes shifts in Firmicutes populations. Front. Cell. Infect. Microbiol. 9, 101 (2019).
Google Scholar
Verduci, E. et al. Phenylketonuric diet negatively impacts on butyrate production. Nutr. Metab. Cardiovasc. Dis. 28, 385–392 (2018).
Google Scholar
Misselwitz, B., Butter, M., Verbeke, K. & Fox, M. R. Update on lactose malabsorption and intolerance: pathogenesis, diagnosis and clinical management. Gut 68, 2080–2091 (2019).
Google Scholar
Azcarate-Peril, M. A. et al. A double-blind, 377-subject randomized study identifies Ruminococcus, Coprococcus, Christensenella, and Collinsella as long-term potential key players in the modulation of the gut microbiome of lactose intolerant individuals by galacto-oligosaccharides. Gut Microbes 13, 1957536 (2021).
Google Scholar
Angima, G., Qu, Y., Park, S. H. & Dallas, D. C. Prebiotic strategies to manage lactose intolerance symptoms. Nutrients 16, 1002 (2024).
Google Scholar
Ang, Q. Y. et al. Ketogenic diets alter the gut microbiome resulting in decreased intestinal Th17 cells. Cell 181, 1263–1275.e16 (2020).
Google Scholar
Olson, C. A. et al. The gut microbiota mediates the anti-seizure effects of the ketogenic diet. Cell 173, 1728–1741.e13 (2018).
Google Scholar
Li, Q., Liang, J., Fu, N., Han, Y. & Qin, J. A ketogenic diet and the treatment of autism spectrum disorder. Front. Pediatr. 9, 650624 (2021).
Google Scholar
Muscogiuri, G. et al. The management of very low-calorie ketogenic diet in obesity outpatient clinic: a practical guide. J. Transl. Med. 17, 356 (2019).
Google Scholar
Fitzpatrick, J. A., Melton, S. L., Yao, C. K., Gibson, P. R. & Halmos, E. P. Dietary management of adults with IBD – the emerging role of dietary therapy. Nat. Rev. Gastroenterol. Hepatol. 19, 652–669 (2022).
Google Scholar
Levine, A. et al. Crohn’s disease exclusion diet plus partial enteral nutrition induces sustained remission in a randomized controlled trial. Gastroenterology 157, 440–450.e8 (2019).
Google Scholar
Armstrong, H., Mander, I., Zhang, Z., Armstrong, D. & Wine, E. Not all fibers are born equal; variable response to dietary fiber subtypes in IBD. Front. Pediatr. 8, 620189 (2020).
Google Scholar
Hume, M. P., Nicolucci, A. C. & Reimer, R. A. Prebiotic supplementation improves appetite control in children with overweight and obesity: a randomized controlled trial. Am. J. Clin. Nutr. 105, 790–799 (2017).
Google Scholar
Birkeland, E. et al. Prebiotic effect of inulin-type fructans on faecal microbiota and short-chain fatty acids in type 2 diabetes: a randomised controlled trial. Eur. J. Nutr. 59, 3325–3338 (2020).
Google Scholar
Limketkai, B. N. et al. Prebiotics for induction and maintenance of remission in inflammatory bowel disease: systematic review and meta-analysis. Inflamm. Bowel Dis. 31, 1220–1230 (2025).
Google Scholar
Talukdar, J. R. et al. The effects of inulin-type fructans on cardiovascular disease risk factors: systematic review and meta-analysis of randomized controlled trials. Am. J. Clin. Nutr. 119, 496–510 (2024).
Google Scholar
Arifuzzaman, M. et al. Inulin fibre promotes microbiota-derived bile acids and type 2 inflammation. Nature 611, 578–584 (2022).
Google Scholar
Yang, J. et al. High soluble fiber promotes colorectal tumorigenesis through modulating gut microbiota and metabolites in mice. Gastroenterology 166, 323–337.e7 (2024).
Google Scholar
Siscovick, D. S. et al. Omega-3 polyunsaturated fatty acid (fish oil) supplementation and the prevention of clinical cardiovascular disease: a Science Advisory from the American Heart Association. Circulation 135, e867–e884 (2017).
Google Scholar
La Rosa, F. et al. The gut-brain axis in Alzheimer’s disease and omega-3. A critical overview of clinical trials. Nutrients 10, 1267 (2018).
Google Scholar
Okburan, G. & Kızıler, S. Human milk oligosaccharides as prebiotics. Pediatr. Neonatol. 64, 231–238 (2023).
Google Scholar
Sanders, M. E., Merenstein, D. J., Reid, G., Gibson, G. R. & Rastall, R. A. Probiotics and prebiotics in intestinal health and disease: from biology to the clinic. Nat. Rev. Gastroenterol. Hepatol. 16, 605–616 (2019).
Google Scholar
Suez, J., Zmora, N., Segal, E. & Elinav, E. The pros, cons, and many unknowns of probiotics. Nat. Med. 25, 716–729 (2019).
Google Scholar
Veiga, P., Suez, J., Derrien, M. & Elinav, E. Moving from probiotics to precision probiotics. Nat. Microbiol. 5, 878–880 (2020).
Google Scholar
Michaudel, C. et al. Rewiring the altered tryptophan metabolism as a novel therapeutic strategy in inflammatory bowel diseases. Gut 72, 1296–1307 (2023).
Google Scholar
Milani, G. P. et al. A systematic review and meta-analysis on nutritional and dietary interventions for the treatment of acute respiratory infection in pediatric patients: An EAACI taskforce. Allergy 79, 1687–1707 (2024).
Google Scholar
Manson, J. E. et al. The Women’s Health Initiative randomized trials and clinical practice: a review. JAMA 331, 1748–1760 (2024).
Google Scholar
Dehghan, M. et al. Associations of fats and carbohydrate intake with cardiovascular disease and mortality in 18 countries from five continents (PURE): a prospective cohort study. Lancet 390, 2050–2062 (2017).
Google Scholar
Zeisel, S. H. Precision (personalized) nutrition: understanding metabolic heterogeneity. Annu. Rev. Food Sci. Technol. 11, 71–92 (2020).
Google Scholar
Lee, B. Y. et al. Research gaps and opportunities in precision nutrition: an NIH workshop report. Am. J. Clin. Nutr. 116, 1877–1900 (2022).
Google Scholar
Hjorth, M. F. et al. Pre-treatment microbial Prevotella-to-Bacteroides ratio, determines body fat loss success during a 6-month randomized controlled diet intervention. Int. J. Obes. 42, 580–583 (2018).
Google Scholar
Christensen, L. et al. Prevotella abundance predicts weight loss success in healthy, overweight adults consuming a whole-grain diet ad libitum: a post hoc analysis of a 6-wk randomized controlled trial. J. Nutr. 149, 2174–2181 (2019).
Google Scholar
Hur, H. J. et al. Beneficial effects of a low-glycemic diet on serum metabolites and gut microbiota in obese women with Prevotella and Bacteriodes enterotypes: a randomized clinical trial. Front. Nutr. 9, 861880 (2022).
Google Scholar
Dao, M. C. et al. Akkermansia muciniphila and improved metabolic health during a dietary intervention in obesity: relationship with gut microbiome richness and ecology. Gut 65, 426–436 (2016).
Google Scholar
Gargari, G. et al. Collinsella aerofaciens as a predictive marker of response to probiotic treatment in non-constipated irritable bowel syndrome. Gut Microbes 16, 2298246 (2024).
Google Scholar
Son, J., Jang, L.-G., Kim, B.-Y., Lee, S. & Park, H. The effect of athletes’ probiotic intake may depend on protein and dietary fiber intake. Nutrients 12, 2947 (2020).
Google Scholar
Wastyk, H. C. et al. Randomized controlled trial demonstrates response to a probiotic intervention for metabolic syndrome that may correspond to diet. Gut Microbes 15, 2178794 (2023).
Google Scholar
Zhang, C. et al. Ecological robustness of the gut microbiota in response to ingestion of transient food-borne microbes. ISME J. 10, 2235–2245 (2016).
Google Scholar
Zmora, N. et al. Personalized gut mucosal colonization resistance to empiric probiotics is associated with unique host and microbiome features. Cell 174, 1388–1405.e21 (2018).
Google Scholar
Nguyen, N. K. et al. Gut microbiota modulation with long-chain corn bran arabinoxylan in adults with overweight and obesity is linked to an individualized temporal increase in fecal propionate. Microbiome 8, 118 (2020).
Google Scholar
Rodriguez, J. et al. Discovery of the gut microbial signature driving the efficacy of prebiotic intervention in obese patients. Gut 69, 1975–1987 (2020).
Google Scholar
Leshem, A., Segal, E. & Elinav, E. The gut microbiome and individual-specific responses to diet. mSystems 5, e00665-20 (2020).
Google Scholar
Wu, W.-K. et al. Characterization of TMAO productivity from carnitine challenge facilitates personalized nutrition and microbiome signatures discovery. Microbiome 8, 162 (2020).
Google Scholar
Cohen, Y., Valdés-Mas, R. & Elinav, E. The role of artificial intelligence in deciphering diet-disease relationships: case studies. Annu. Rev. Nutr. 43, 225–250 (2023).
Google Scholar
Zeevi, D. et al. Personalized nutrition by prediction of glycemic responses. Cell 163, 1079–1094 (2015).
Google Scholar
Rein, M. et al. Effects of personalized diets by prediction of glycemic responses on glycemic control and metabolic health in newly diagnosed T2DM: a randomized dietary intervention pilot trial. BMC Med. 20, 56 (2022).
Google Scholar
Ben-Yacov, O. et al. Personalized postprandial glucose response-targeting diet versus Mediterranean diet for glycemic control in prediabetes. Diabetes Care 44, 1980–1991 (2021).
Google Scholar
Berry, S. E. et al. Human postprandial responses to food and potential for precision nutrition. Nat. Med. 26, 964–973 (2020).
Google Scholar
Valdés-Mas, R. et al. Metagenome-informed metaproteomics of the human gut microbiome, host, and dietary exposome uncovers signatures of health and inflammatory bowel disease. Cell 188, 1062–1083.e36 (2025).
Google Scholar
link





:max_bytes(150000):strip_icc()/Health-GettyImages-1413749294-1e0959850fd24395b36963b6b3f2e109.jpg)
