Variations in oral health outcomes and mycobiome composition among COVID-19 convalescents
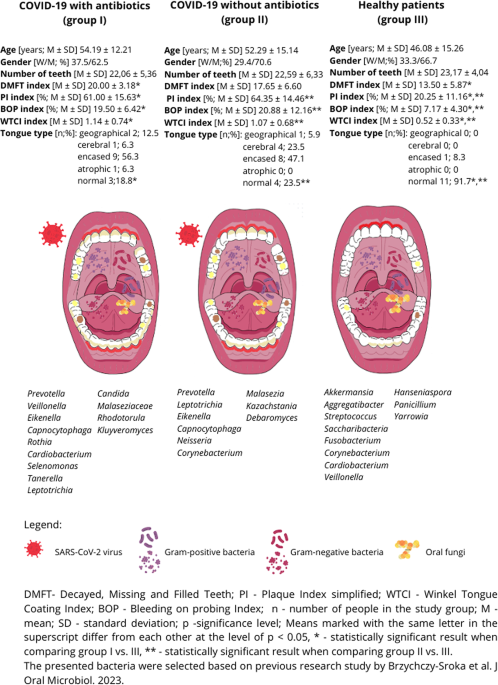
The condition of the oral cavity and its resident microbiota are interrelated elements, the balance or disruption of which may lead to the onset of various diseases. Therefore, maintaining the health of the oral cavity—including the soft tissues and teeth—is crucial for the overall well-being of the human body. Earlier research has demonstrated that periodontal pockets create a conducive environment for the development of microbiota abundant in bacterial and viral species, which may invade the tissues through the frequently ulcerated pocket epithelium. This could enable the SARS-CoV-2 virus to enter the body either directly through these damaged epithelial layers or indirectly by enhancing the expression of angiotensin-converting enzyme 2 (ACE-2) receptors27. In the study we conducted, people who were infected with SARS-CoV-2 had significantly higher values of dental indicators (DMFT, PI, BOP, WTCI) than those who were not infected with the virus. Elevated DMFT, PI, and BOP indices among COVID-19 convalescents reflect chronic structural changes (such as carious lesions and loss of tooth attachment) and increased periodontal inflammation, likely attributable to the prolonged disease course. Our findings align with previous findings that local gingival inflammation may promote early and long-term SARS-CoV-2 colonization8. However, in contrast to previous reports8, we did not observe the increased prevalence of geographic tongue as a characteristic manifestations of COVID-19. Additionally, we did not identify any statistically significant correlations between the morphologically distinct tongue types and the types of fungi in any of the study group. A critical aspect of our study is that it was conducted during the COVID-19 pandemic, requiring strict adherence to hospital sanitary protocols. Performing dental examinations and collecting samples under these conditions posed a significant challenge, as ensuring patient and examiner safety while maintaining the highest standards of medical practice was of utmost importance. Opposite to our research, Larvine et al. based on results self-reported by patients such as bleeding gums, painful gums, and loose teeth which were used as proxies for diagnosing periodontal disease. Larvine et al. assumed that painful and bleeding gums were associated with mild to moderate periodontal disease, while loose teeth indicated severe periodontal disease. In contrast, participants who did not report these symptoms were classified as the control group, representing individuals without periodontal disease28. Another approach to assessing oral hygiene and health during the SARS-CoV-2 pandemic was to perform radiological examinations29. Among the teams conducting dental examinations on COVID-19 patients, DMFT, PI, and BOP indexes were comparable to ours. In the study by Costa et al.30 (DMFT index = 18), which assessed oral health status and adverse COVID-19 outcomes in hospitalized patients, we gained similar findings (DMFT index = 20.00 ± 3.18 for group I and DMFT = 17.65 ± 6.60 for group II, respectively). Moreover, in Anand P et al. research the participants with COVID-19 had significantly higher mean values of plaque scores (0.77 ± 0.50 vs. 0.29 ± 0.30, respectively, p < 0.001) and gingival bleeding scores (0.62 ± 0.24 vs. 0.29 ± 0.20, respectively; p < 0.001) compared to the controls, which is similar to our findings for I and II group31. Based on the observations mentioned above, it can be concluded that there was an association between COVID-19 and periodontitis severity. Gingival bleeding and dental plaque accumulation were also more frequent among COVID-19 patients.
The statistically significant differences in alpha diversity observed between COVID-19 convalescents who received antibiotic treatment during hospitalization and those who recovered from COVID-19 without antibiotic therapy, as well as individuals who were not infected with the virus, were unexpected. We hypothesize that the observed outcomes can be attributed to the significant interindividual variability in microbiota composition, which varies widely among individual convalescents and volunteers included in our research (Fig. 4B). In contrast to our findings, three other studies found no significant differences in alpha diversity among the patients studied6,10,18 although it should be noted that each of these studies analysed different types of biological samples, including mouthwashes (Gupta et al.), throat swabs (Wei N. et al.) and tongue coating samples (Xiaobo Hu et al.). However, one of the study included patients during SARS-CoV-2-infection and healthy controls who were not treated with antibiotics18. In the study by Wei et al., recovered COVID-19 patients were examined 1 year after infection but were also not treated with antibiotics for 6 months prior to the examination6. The study analyzed 71 COVID-19 patients, 22 recovered patients, 36 suspected cases, 36 recovered suspected cases, and 132 controls10.
In turn, our study did not observe a clearly distinct mycobiota composition for the individual groups measured with the beta diversity, which indicates similarity between them (Fig. 1S). We hypothesize that the absence of significant differences among our patients’ mycobiota could be attributed to the mild nature of their COVID-19 cases. The relatively mild severity of the disease in these patients may not have been a sufficient factor to induce notable alterations in the mycobiota. Results similar to ours were obtained by Wei et al. studying differences in the oral fungal community of throat swabs in recovered COVID-19 and healthy controls. The above observations are explained by some authors as being due to the significantly lower variability of fungi compared to the higher variability of bacteria comunity6.
Nevertheless, we have observed the fungal quantitative composition in COVID-19 recovered patients, both those who received antibiotics during hospitalization and those who did not, differ from that of healthy individuals. The phyla Ascomycota and Basidiomycota were predominant in both COVID-19 patients and healthy individuals, but the percentage share of Ascomycota to Basidiomycota was highest in patients who took antibiotics during COVID-19 treatment. This can be explained, among other things, by the fact that the high prevalence of the Candida genus, which not only exhibited the highest relative abundance in Group I but was also identified as a key differentiating taxon in the Linear Discriminant Analysis (LDA).
The Candida genus is well known as the common opportunistic fungal genus in the oral cavity32. The overgrowth of Candida can lead to thrush or invasive candidiasis and candidemia33 and even promoting oral cancer34 and colon cancer35. Candida pathogenicity can be promoted via immunosuppression, the prolonged use of antimicrobials and corticosteroids, and direct cell damage by SARS-CoV-233. In healthy people bacteria in the oral cavity colonize the mucus membrane and can limit the invasion of fungi and viruses36. Our previous studies on the oral microbiome in patients who recovered from COVID-19 showed that SARS-CoV-2 colonization and the administration of antibacterial drugs limited bacterial growth11. It might contributed to the loss of mechanisms inhibiting the development of opportunistic fungi and initiated Candida invasion what we observed in our present research. Genus Candida may destroying immune cells and promote inflammation itself having virulence factors such as production of phospholipases, proteases, hemolysin, candidalysin, and biofilm formation responsible for adherence and invasion37. The overrepresentation of Candida genus in both the I and II groups compared to the control group, as demonstrated by percentage composition and the LefSE method, suggests a potential risk for the development of oral candidiasis in these patients. Previous studies have reported the occurrence of candidiasis in COVID-19 patients, particularly in those who experienced severe cases or were immunocompromised16,38. However, our study cohort consisted of patients who were in good health at the time of examination and sampling, which was conducted on the day of discharge following hospitalization for COVID-19. These patients did not exhibit impaired immunity and were not admitted to the intensive care unit, conditions that are often associated with secondary infections, including oral candidiasis, post-COVID-1916. In the study by Singulani et al. fungal infections were diagnosed after more than two weeks of hospitalization16. Therefore, fungal infections often develop later, progress more slowly than bacterial infections, and are influenced by various risk factors, for example antibiotic administration. Our patients were mainly treated by ceftriaxone as the many other patients hospitalized from COVID-19 during the first phase of the SARS-CoV-2 pandemic39. Maeda et al. used azithromycin and levofloxacin (narrow-spectrum antibiotics) for the mild COVID-19 group, and meropenem, and tazobactam/piperacillin (broad-spectrum antibiotics), for the severe group infected by SARS-CoV-2 virus. They found that the composition of Candida species and the fungal beta diversity showed no significant difference between the severe and mild study groups. Moreover, Maeda et al. reported the alpha diversity of mycobiota in the severe group was reduced, but no difference was observed between the groups with and without meropenem. They stated the effect of antibiotic use on mycobiota alterations is not significant in the gut40. We did not find any studies describing this phenomenon in the oral cavity. Another factor considered to be conducive to the occurrence of oral candydosis are dental prostheses, which is confirmed by the results of studies reporting the occurrence of denture stomatitis in 60% of patients with COVID-19 using dentures, most often caused by Candida albicans41. In our study, only two patients reported the use of dentures. These patients had a characteristic quantitative composition of fungi, in which we observed an overrepresentation of Candida genus. Surprisingly, in the saliva from patients recovered from COVID-19 wearing the dentures were fungi from the Debaromyces genus. Previous studies reported an increase in the number of Debaromyces spp. in inflammatory bowel diseases in children and mouse model42,43. Fungi from the genus Debaromyces can maintain inflammation, which undoubtedly occurs in patients wearing dentures and suffering from COVID-19, which we confirmed by the increased dental coefficients observed in these patients. However, the observations we made based on these two patients should be repeated in a larger study group.
Going deeper, we analyzed the mycobiome in parallel with the bacteriobiome from our previous research11 which will help understand synergism and antagonism between genera of fungal and bacterial kingdoms, which may promote or prevent COVID-19 infection and outcomes. Microorganisms such as bacteria and fungi release various signaling molecules and chemicals that can either inhibit or support each other’s growth and survival, thereby influencing the host’s immune response and overall health4,10. A well-documented fungal-bacterial interaction in the oral microbiome is Candida albicans colonization linked to Streptococcus spp. and Porphyromonas gingivalis, which alters microbial composition, worsens carious lesions, and accelerates tooth demineralization44,45. Sztukowska et al. described that the InlJ protein from P. gingivalis binds to the Als3 protein on C. albicans hives, which promotes the formation of common biofilms and potentially increases the virulence of these microorganisms45. Bartnicka et al. found that in biofilms C. albicans creates a protective environment for P. gingivalis under aerobic conditions, which is possible due to the expression of adhesion proteins such as Als3 and aspartyl proteases Sap6 and Sap946. In our study, Candida genus was strongly negatively correlated with Porphyromonas, in group I. Our findings suggest that Candida in saliva may act as a limiting factor for Porphyromonas colonization. One possible explanation is the impact of antibiotic treatment administered during the hospitalization of COVID-19 patients, which may have affected Porphyromonas populations. Additionally, in saliva, the biofilm formed by Candida and Porphyromonas is less structured compared to dental plaque. As a result, an increase in Candida may exert an opposite effect, restricting the growth of P. gingivalis rather than promoting its survival, as observed in plaque-associated biofilms. Another potential mechanism at play in convalescent patients involves the immune response triggered by Candida. This fungal species can stimulate host immunity, leading to the production of defensins, cytokines, and other antimicrobial factors that may suppress P. gingivalis proliferation. Furthermore, Candida produces various metabolites, including organic acids and hydrogen peroxide, which could influence the growth and viability of anaerobic bacteria such as Porphyromonas. Furthermore, heme competition may explain the antagonism between Candida albicans and Porphyromonas gingivalis in saliva. Both microorganisms rely on heme for growth, so Candida may outcompete Porphyromonas limiting its proliferation. Unlike in subgingival biofilms, where C. albicans enhances P. gingivalis virulence under heme scarcity, in saliva, weaker biofilm formation and transient interactions may favor Candida genus dominance47. Our findings, along with those of other researchers, highlight the context-dependent nature of the Candida–Porphyromonas relationship, underscoring the need for further investigation. In the group of COVID-19 convalescents who received antibiotic treatment during hospitalization, we also observed a strong positive correlation between fungi of the Yarrowia genus and bacteria of the Aggregatibacter genus. Unlike Candida, the presence of Yarrowia in the oral cavity is poorly documented, and its pathogenic potential remains largely unknown48. We suspect that in this case, Yarrowia genus might serve as a physical scaffold for Aggregatibacter, facilitating its adhesion to oral surfaces. In oral biofilms, interactions between bacteria and fungi often contribute to mutual protection against the host immune system. Our other results, which show weaker correlations with various microorganisms (Table 2), suggest that the biological roles of Yarrowia are not uniform and may depend on the surrounding environment as well as the fungal and bacterial communities with which the Yarrowia genus coexists.
We discovered that Malassezia was overrepresented in COVID-19 convalescents who did not receive antibiotics during hospitalization (group II). Dupuy et al. identified the Malassezia genus as a dominant fungal genus in the oral microbiome. Moreover, their research demonstrates that Malassezia is consistently present in healthy individuals, suggesting a potential role in oral homeostasis. Notably, their findings align closely with our results, further supporting the significance of Malassezia in the oral environment49. Additionally, Malassezia has been implicated in cases of systemic inflammation during the progression of COVID-1913.
At the same time, we noticed the overrepresentaion of molds such as Aspergillus and Penicillium in the saliva of our control group (Fig. 4C,D). Furthermore, we showed a strong positive correlation of the genus Aspergillus with the genus Cladosporium and Amyloporia and Cladosporium. For example, Aspergillus and Cladosporium have previously been identified as genus-level constituents of the oral mycobiome32,49. Due to their ubiquitous nature, the presence of these fungi in the oral cavities of healthy individuals was interpreted as most likely of environmental origin, from food and breathing49. Wei et al.6 reported results similar to ours regarding the Aspergillus genus, observing a prevalence of 1.34% in COVID-19 recovered patients compared to 2.49% in healthy controls. We would also like to point out that we have demonstrated fungi in individual patients that are not yet well characterised as residents or oral pathogens. Nevertheless, they were present in the saliva of the patients we examined or of the control subjects. For example, we noticed characteristic overrepresentation of Kazachstania genus due to the high percentage of this fungus in two patients in particular. The genus Kazachstania is rarely described as a human pathogen to date50. In our patients, one of them when the Kazachstania genus was present, there was no Candida genus (no. 2815, Fig. 3B), but in the second one, there was the co-presence of Kazachstania and Candida genera. It should be investigated in the larger study group in the future. In the study designed by Kralova et al. Kazachstania heterogenica var. weizmannii was discovered as murine commensal that antagonizes C. albicans colonization and significantly reduces its abundance in animal model51. This suggests that Kazachstania may play a protective role in reducing Candida proliferation, potentially mitigating fungal overgrowth in the oral cavity.
We made significant efforts to elucidate the alterations and roles of oral fungi in patients who have recovered from COVID-19; however, certain limitations remain in this study. One of the limitations of our research was the challenges associated with existing fungal taxonomy databases. Enhancing these databases through a more comprehensive classification of taxa would significantly aid in advancing our understanding of oral fungal ecology in mycobiome research. Another aspect to consider is that our findings are based on samples collected from a specific geographic area, so the observed changes in composition might not be representative of COVID-19 patients from different regions or countries. Similar limitations were noted in the studies by Rizzello et al.13.
Additional large-scale prospective studies are necessary to compare the mycobiome of COVID-19 patients with that of contemporary healthy individuals. Such research could provide further validation of our findings and contribute to corroborating existing data. Despite some shortcomings of our study, we achieved our goal, which was to provide a broader perspective on SARS-CoV-2 infection and to evaluate the interaction between oral health and microbiota, with a particular focus on the mycobiome.
link





:max_bytes(150000):strip_icc()/Health-GettyImages-1413749294-1e0959850fd24395b36963b6b3f2e109.jpg)